The Mysterious World of Atoms
A lot can be said about the concept of "only want til atom." In reality, most atoms don't want to have 8 atoms, as this is just a simplification taught in school. Many atoms want more, and the actual number is 2, 8, 18, 32, 32, and so on. However, some special atoms don't want a full outer shell.
Understanding the Octet Rule

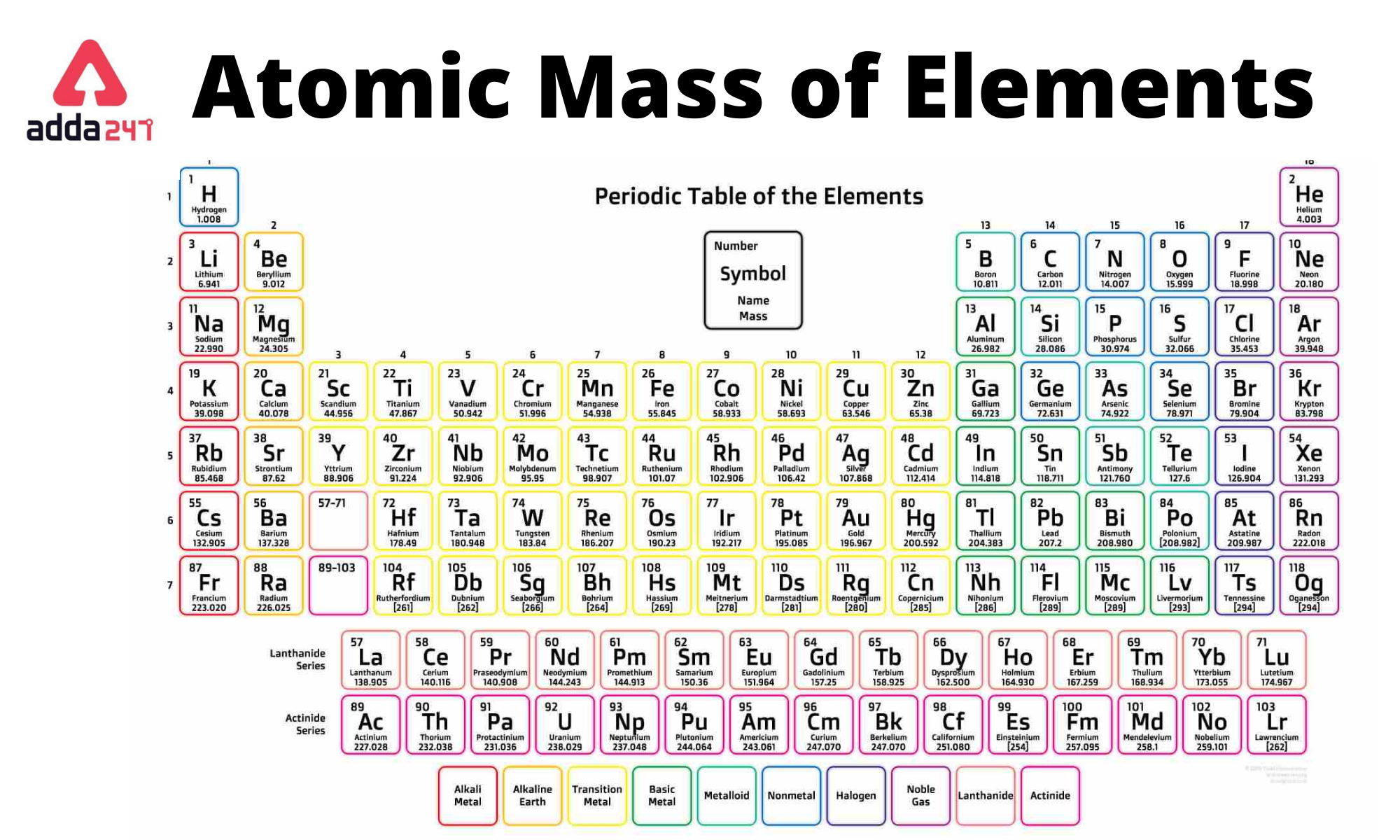
Atoms are the building blocks of matter, and they play a crucial role in forming chemical bonds. The octet rule is a fundamental concept in chemistry that explains how atoms tend to gain, lose, or share electrons to achieve a full outer shell, which consists of 8 electrons. This rule is based on the idea that atoms want to be in a stable state and reach a full outer shell to achieve this stability.
- Atomic Number:** The atomic number of an atom determines the number of electrons it has in its outer shell. Atoms with a low atomic number tend to lose electrons, while those with a high atomic number tend to gain electrons.
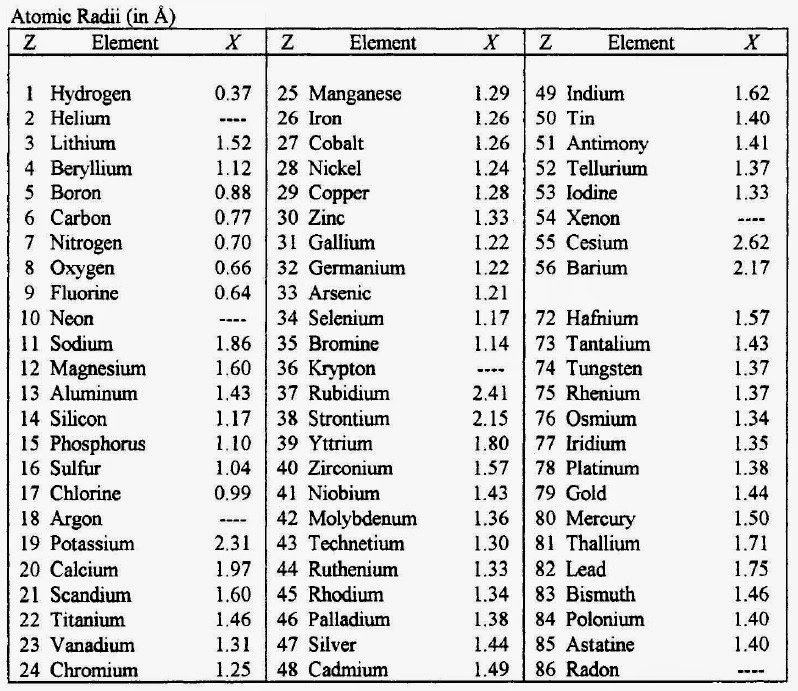
- Isotopes:** Atoms can have different numbers of neutrons, which affects their mass but not their chemical properties. The isotopes of an element have the same number of protons but differ in the number of neutrons.
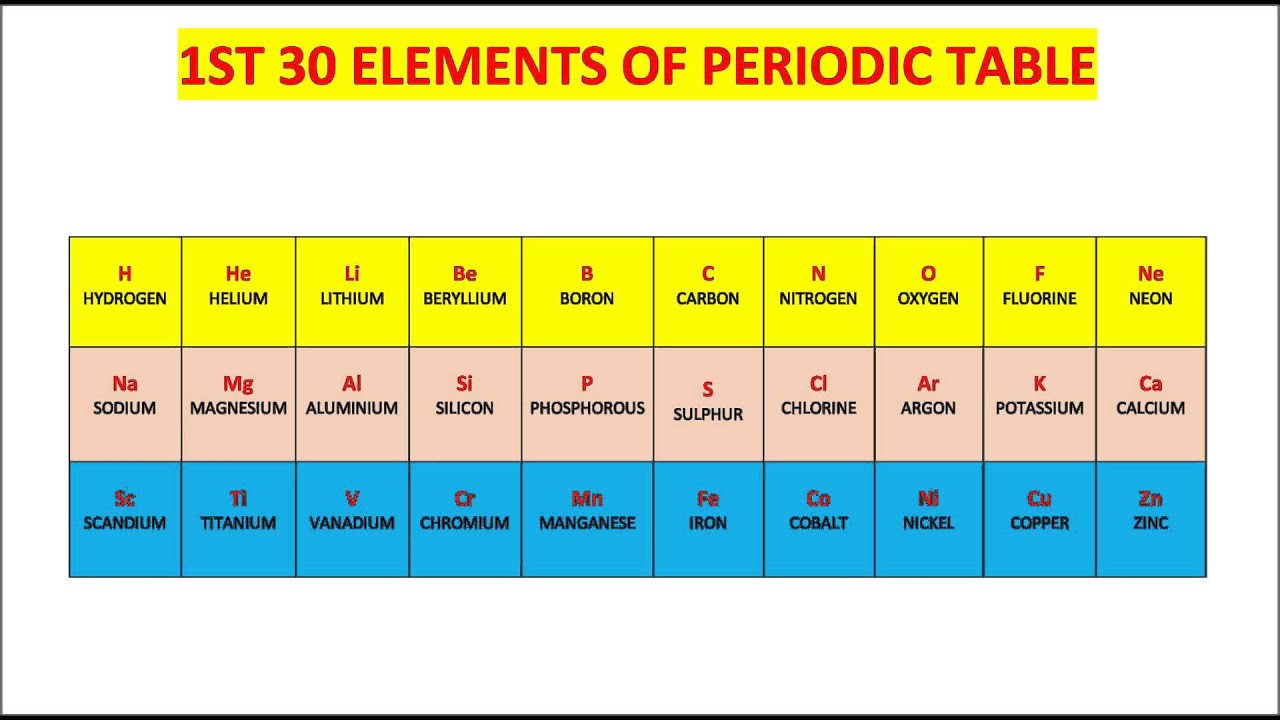
- The Periodic Table:** The periodic table organizes elements based on their atomic number and electron configuration. Elements with similar properties are grouped together in the periodic table.














![Eminem - Till I Collapse [HD] - YouTube Eminem - Till I Collapse [HD] - YouTube](https://i.pinimg.com/originals/b7/b3/0d/b7b30dd1732e355ee7a9a49e5ea4f7f6.jpg)









