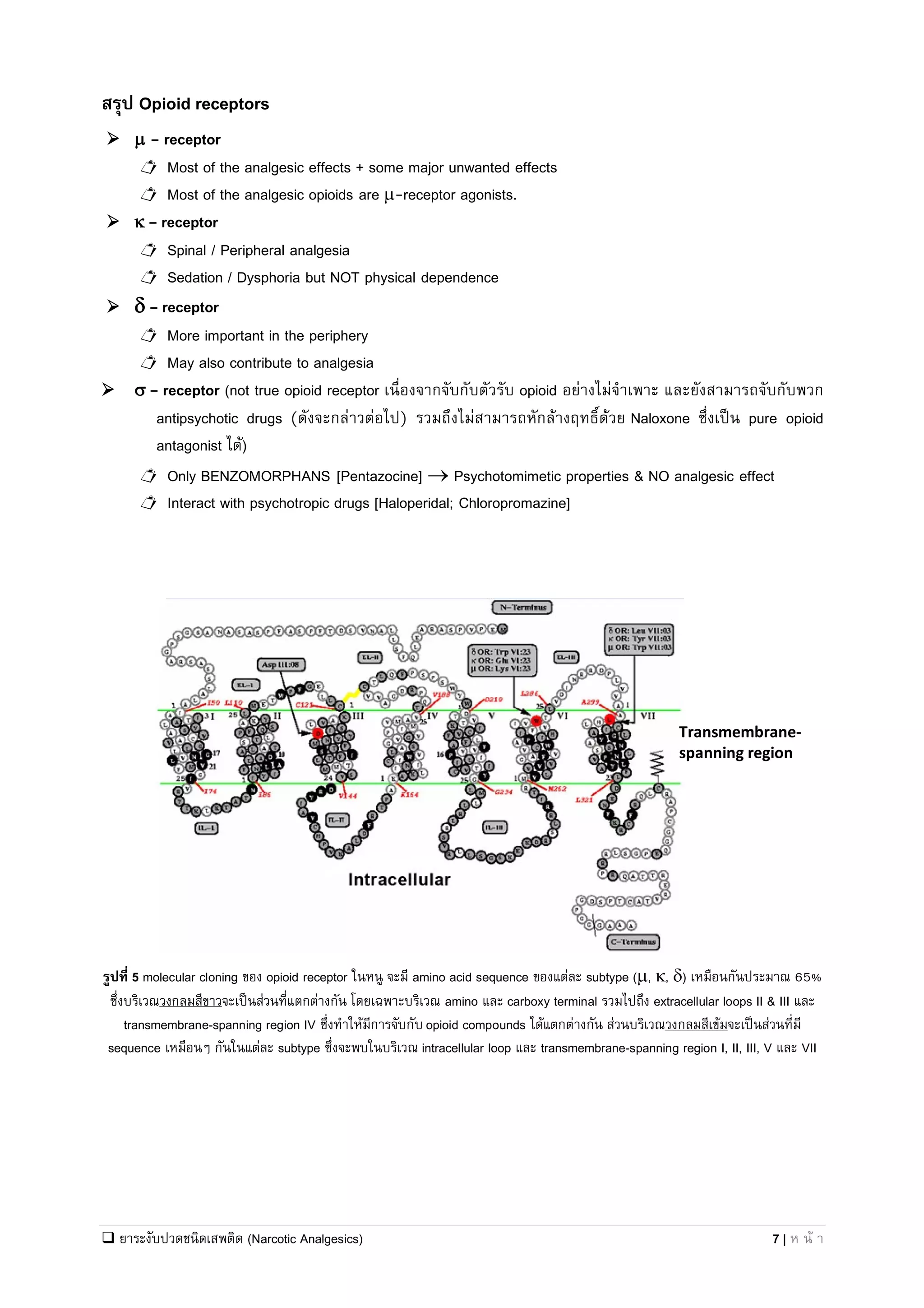
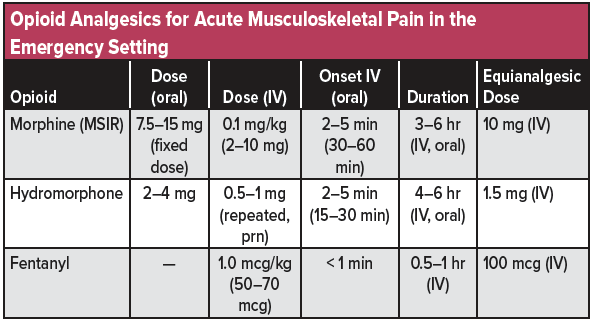
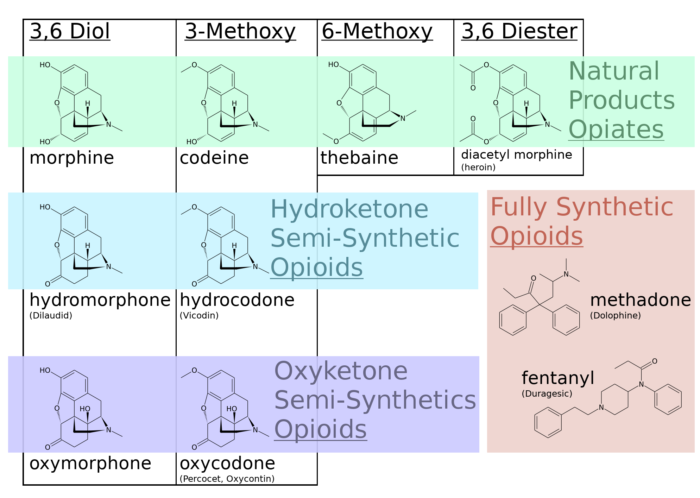
Regulatory Importance of Casting a Regulatory Net Around Pil Volta Opioid Analgesic
As the opioid epidemic continues to ravage communities across the globe, regulatory agencies and healthcare professionals are working together to cast a net around the use of opioid analgesics, ensuring that these powerful pain relievers are used safely and responsibly. In this article, we will explore the regulatory importance of casting a regulatory net around pil volta opioid analgesic, highlighting the efforts of regulatory agencies, healthcare professionals, and industry stakeholders in mitigating the risks associated with opioid use.
The Opioid Epidemic: A Public Health Crisis
The opioid epidemic is a public health crisis that has been exacerbated by the overprescription of opioid analgesics. According to the Centers for Disease Control and Prevention (CDC), more than 130 people in the United States die every day from opioid overdose, with the majority of these deaths linked to prescription opioids. The situation is not contained to the United States, with many other countries experiencing similar issues with opioid misuse and addiction.
Regulatory Agencies Step in to Mitigate Risks
Regulatory agencies, including the U.S. Food and Drug Administration (FDA), have taken various measures to address the opioid crisis. In 2016, the FDA introduced the Opioid Analgesic Risk Evaluation and Mitigation Strategy (OA REMS) to reduce the risk of abuse, misuse, addiction, overdose, and death associated with opioid use. The OA REMS requires manufacturers to implement educational programs for healthcare professionals and patients, as well as to ensure that opioid analgesics are prescribed and used safely.